FDA Advisory Panel Recommends HPV Vaccine
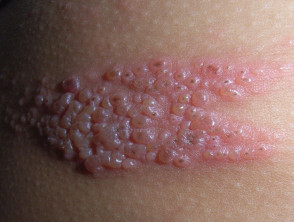
An FDA advisory committee on Thursday voted 13-0 that Merck's experimental human papillomavirus vaccine Gardasil is safe and effective for girls and women ages nine to 26, which effectively recommends that the agency approve the vaccine for sale and marketing to the age group, the Wall Street Journal reports (Corbett Dooren, Wall Street Journal, 5/19). Merck in October 2005 announced that Gardasil in clinical trials was 100% effective in preventing infection with HPV strains 16 and 18, which together cause about 70% of cervical cancer cases (Kaiser Daily Women's Health Policy Report, 5/18). The trials showed the vaccine -- given in three injections over six months -- also was about 99% effective in preventing HPV strains 6 and 11, which together cause about 90% of genital wart cases (Allday, San Francisco Chronicle, 5/19).
Merck on Thursday also asked the advisory committee to recommend approval for use of Gardasil in boys ages nine to 15, even though studies on the age group are not complete. Committee members did not vote on the request (Ginsberg, Philadelphia Inquirer, 5/19). Monica Farley, an infectious disease specialist at Emory University School of Medicine and panel chair, said FDA had not requested the advisory panel to consider the safety and efficacy of approving the vaccine for boys (Henderson, Boston Globe, 5/19).
FDA in a document released on Thursday had asked the panel to question whether congenital abnormalities among five infants born to women who gave birth within a month of being vaccinated with Gardasil were linked to the vaccine (Kaiser Daily Women's Health Policy Report, 5/18). Nancy Miller, FDA medical reviewer, told the panel that there is no pattern in the abnormalities among the five infants, which suggests that there was no link to Gardasil (Wall Street Journal, 5/19).
Although FDA is not required to agree with the advisory panel's recommendations, it often follows them. Merck in February announced that FDA had granted priority review for Gardasil, which means the agency has six months from the time of the company's application to make a decision on the vaccine's approval.
The six-month period ends June 8 (Kaiser Daily Women's Health Policy Report, 5/18). Merck Executive Director of Medical Affairs Rick Haupt said the company is prepared to make Gardasil available "within weeks" of FDA's decision (Graham, Chicago Tribune, 5/19).
Who Would Receive Gardasil?
If Merck's application for Gardasil receives FDA approval, it also must be examined by CDC's Advisory Committee on Immunization Practices, which drafts recommendations and schedules for the administration of vaccines in the U.S. (Kaiser Daily Women's Health Policy Report, 5/18). ACIP's HPV vaccine group has recommended giving the vaccine to girls ages 11 and 12, the AP/Forbes reports. The 15-member ACIP also will consider recommending Gardasil for girls and women ages 13 to 26 (Bridges, AP/Forbes, 5/18). However, CDC only can recommend who should get the vaccine, and any vaccination requirements would be determined by states, the Los Angeles Times reports (Maugh/Gellene, Los Angeles Times, 5/19).
Some conservative groups initially were concerned that FDA approval of the vaccine could be seen by some girls as an endorsement of sex, according to the Baltimore Sun (Rockoff, Baltimore Sun, 5/19). However, Merck "quietly dispatched representatives" to meet with leaders of those groups to explain the scientific rationale for mass immunization at an early age, "a move that appears to have paid off ... [f]or now," the Dallas Morning News reports (Beil, Dallas Morning News, 5/19). No group spoke in opposition to the vaccine's approval at Thursday's meeting, but many members of conservative groups hinted that they would not want their daughters vaccinated, the Times reports. "We welcome the vaccine," Pete Sprigg of the Family Research Council said. He added that "decisions about sexual health are tied up in personal values, and parents should have the right to transmit personal values to their children" (Los Angeles Times, 5/19).
Other Issues
FDA advisory panel member Bruce Gellin said the vaccine could "prevent the bulk" of HPV infections, "but not all of them," adding, "Even if you're vaccinated, it doesn't mean you're absolutely prevented from developing these infections." Merck said it would market Gardasil as a complement to Pap tests. "This company is absolutely committed to cervical cancer screening," Eliav Barr, Merck senior director of clinical vaccine and biologics research, said, adding, "This vaccine is not a replacement for cervical cancer screening" (Boston Globe, 5/19). Merck also is advising young people to "avoid sexual contact of any type," Haupt said, adding, "[C]ounseling adolescents (not to engage in any type of sex) is the only way to prevent infection" (Howard Price, Washington Times, 5/19).
In addition, Gardasil is expected to cost about $300 to $500 per vaccine, which could prevent access to the vaccine to millions of people worldwide. Merck said it is talking to the Bill & Melinda Gates Foundation about how to provide Gardasil in developing countries, according to the Chronicle (San Francisco Chronicle, 5/19). According to the Morning News, some public health experts see the rollout of Gardasil as a "kind of a dress rehearsal" for any potential HIV/AIDS vaccine. "This is something we're all going to be watching," Ruth Faden, director of the Phoebe R. Berman Bioethics Institute at Johns Hopkins Bloomberg School of Public Health, said (Dallas Morning News, 5/19).
KaiserNetworkOrg
May 19, 2006