Human Papillomavirus DNA Testing
The National Cervical Screening Program recommends Pap smears be used as the primary method for screening for cervical abnormalities and that testing for high risk types of HPV DNA testing be used for monitoring as a ‘test of cure’, for women following treatment for high-grade intra epithelial disease of the cervix.
Key Points
- The National Cervical Screening Program (NCSP) recommends that women aged 18-69 years, who have ever been sexually active 2, should have cervical screening by Pap smears every two years. This includes women who have been immunised with the HPV vaccine.
- The National Health and Medical Research Council (NHMRC) Screening to prevent cervical cancer: guidelines for the management of asymptomatic women with screen-detected abnormalities recommend high risk HPV DNA testing for women who have been treated for biopsy confirmed high grade cervical abnormalities to ensure the HPV infection linked with that occurrence of high grade cervical abnormality has resolved. HPV DNA testing for this purpose is subsidised by a Medicare rebate.
- Currently, HPV DNA testing outside the NHMRC Guidelines is not recommended or covered by Medicare.
- There is no role for HPV DNA testing before giving the HPV vaccine.
Rationale
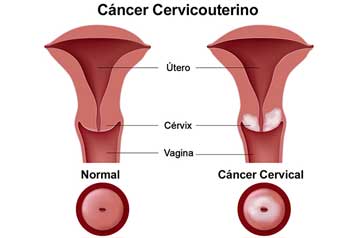
Cervical cancers are associated with persistent infection with oncogenic human papillomavirus (high risk HPV). A high risk HPV DNA test can detect the presence of oncogenic strains of HPV in cervical cells.If a woman requests HPV DNA testing it is important to ensure that she understands the meaning of the test. It will only detect an HPV infection that is present at the time of the test. It does not detect infection that has occurred in the past and has been cleared.
It is also important that a woman requesting HPV DNA testing realises that most infection, even with high risk strains of HPV, will usually resolve within 12-18 months with no treatment and that the HPV DNA test cannot predict the small proportion of such infections which will not clear spontaneously.
HPV DNA testing does not detect abnormal cell changes in the cervix as the Pap smear does. A woman who tests positive for HPV will still require a Pap smear to check for possible cervical abnormalities.
Well women who have had an HPV DNA test outside the recommended guidelines should continue to have regular Pap smears irrespective of their HPV DNA test result.
The Pap smear remains the most effective method for the early detection of cervical cancer and its precursors. The NCSP recommends routine screening with Pap smears every two years for women between the ages of 18 and 69 years who have ever been sexually active.
There is no role for HPV DNA testing of women who are contemplating receiving the HPV vaccine, in determining whether they are likely to benefit from vaccination. HPV DNA testing detects only current infection and will not identify whether the woman has had previous HPV infection. In addition, readily available tests do not specifically identify whether current or past infection is due to a vaccine type HPV. The result of the test would therefore not alter the recommendation concerning the woman and HPV vaccine.
There is a Medicare rebate available for testing for high risk HPV as a ‘test of cure’ for women previously treated for highgrade intraepithelial lesions (HSIL). A woman treated for HSIL requires a colposcopy and cervical cytology at 4-6 months after treatment. Cervical cytology and high risk HPV DNA testing should be carried out 12 months after treatment has occurred and then every 12 months until the woman has tested negative for both tests on two consecutive occasions. This is in line with recommendations made in the NHMRC Screening to prevent cervical cancer: guidelines for the management of asymptomatic women with screen-detected abnormalities.
The Medical Services Advisory Committee (MSAC) has reviewed HPV DNA testing on two previous occasions. HPV DNA testing for triaging of women with equivocal screening results was reviewed in 2002 3 and HPV DNA testing for cervical screening either alone or as an adjunct to cytology was reviewed in 2003. 4 On both occasions, MSAC found there was insufficient evidence to support the use of the technology in the NCSP.
New evidence related to the use of HPV DNA testing for cervical screening has become available since these earlier reviews. MSAC is currently assessing a new application for HPV DNA testing as a triage test for women with Pap smear detected lowgrade squamous intra epithelial lesions. The role of HPV DNA testing in the NCSP will be reviewed once the results of the MSAC assessment are known.
Addendum added 14 May 2011
The above review was completed by MSAC in 2009. MSAC assessed HPV triage testing for women with possible or definite lowgrade squamous intraepithelial lesions. MSAC found that compared with repeat recall cytology at one year, HPV triage testing is safe and effective but not cost effective in the Australian setting at the current price.5
Endorsed by the Australian Population Health Principal Committee at its 6 February 2009 meeting.
Source
Human papillomavirus (hpv) dna testing
Australian Government Department of Health and Ageing
Related Topics
-
Screening for Cervical Cancer: U.S. Preventive Services Task Force Recommendation Statement. 19 June 2012
Ann Intern Med. 19 June 2012;156(12):880-891 -
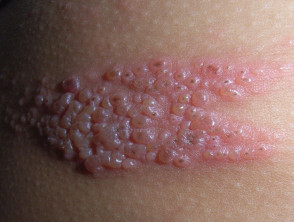
Genital Warts Treatment Guidelines 2010.
Sexually Transmitted Diseases Treatment Guidelines, 2010
Centers for Disease Control and Prevention -
Prevención y Detección Oportuna del Cáncer Cérvico Uterino
en el Primer Nivel de Atención. México. Secretaria de Salud, 2008 -
Canadian Consensus Guidelines in HPV. Society of Obstetricians and
Gynaecologists of Canada. August 2007. Archivo PDF 1.031 KB -
Human Papillomavirus DNA Testing. Australian Population Health Principal Committee. February 2009
-
Human Papillomavirus